For a number of cosmetic products, inhalation is also a potential exposure route. Below there are some examples of cosmetic products in which inhalation exposure is relevant under the normal and foreseeable use of this product category:
- Nail polishes that contain substances (e.g., solvents) with a low boiling point and high vapour pressure.
- Spray products (both pump and propellant spray).
- Cosmetic powders (e.g., face powder).
For all these products, apart from the primary dermal exposure, the exposure via inhalation has to be evaluated. Specifically, both systemic and local adverse health effects on the lungs/airways should be investigated in the risk assessment. For the appropriate evaluation of the toxicity via inhalation, knowledge of the hazard profile of the cosmetic ingredients, their concentrations in the final product, and the likely exposure scenario of the final product is needed to calculate the margins of safety.
The inhalation of respirable droplets/particles from cosmetic products, including pump and propellant sprays, is likely to be very small, even negligible, compared with dermal contact and other exposure routes associated with the use of these products. However, even such small inhalation exposures may be significant for an ingredient that has the potential to act as a potent systemic or local respiratory tract toxicant or to accumulate in the body. Typically, <1% of the airborne droplets/particles released from pump sprays are in the range considered to be respirable (i.e., (aerodynamic equivalent diameters) dae <10 μm). In comparison, the median dae of the airborne droplets/particles of propellant sprays range from 25 μm to 50 μm. Usually, 1% to 2.5% but no more than 5% of the droplets/particles emitted from propellant hair sprays are within the respirable range.
In the absence of a realistic assessment of droplet size for the final product, a conservative exposure calculation for secondary inhalation should be considered.
The safety assessor can primarily use the simpler and more conservative method (i.e., the "1-box model") as described in the 11th Version of SCCS Notes of Guidance, and then use the more refined method (i.e., the "2-box model") if, for example, the safety margin is considered to be too low at the use of the more conservative method.
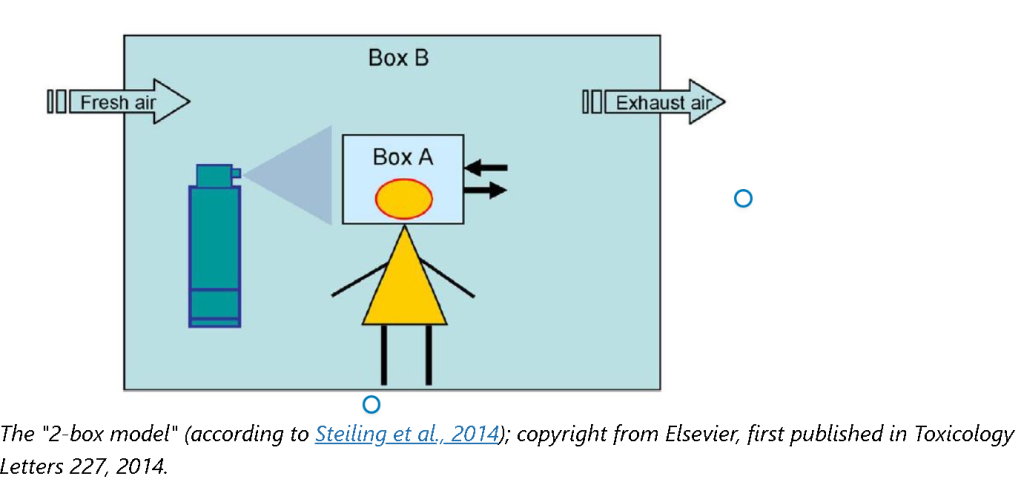
On the one hand, for the "1-box model", the assumption is that the spray is instantly released and distributed in a Box A around the head. On the other hand, the "2-box model" is the worst-case scenario as this takes into account the duration of exposure in both near-field around the head (Box A) and far-field (e.g., bathroom), (Box B). The "2-box model" considers the dilution of the substance over time. There, the aerosol is present for exposure over a defined time, after which the full amount of aerosol in the first box (Box A) is transferred to a larger second Box B.
In order to demonstrate that a cosmetic product complies with Article 3 (safety) of the Cosmetics Regulation, the Responsible Person shall ensure that the product has undergone a safety assessment and is in accordance with Annex I to the Regulation taking into account all routes of exposure occurring during the normal and foreseeable use of cosmetic products.
