On 7 November 2023, Cosmetics Europe published a guidance called "Guidelines on the fragrance allergens requirements" to further explain Regulation (EU) 2023/1545, which amends Regulation (EC) No 1223/2009 concerning the labeling of fragrance allergens in cosmetic products.
In particular, the guidance has been developed to support the cosmetics industry to deal with complex cases that may result from the implementation of the new requirements for the labeling of allergens. In addition, the annex to the guidance helps global actors understand the new requirements and the potential impact these may have in their countries and internationally. The new labeling requirements aim to provide consumers with information about the presence of allergens in cosmetic products. Currently, 24 fragrance allergens must be listed individually by law. The new Regulation expands this list to 81 fragrance allergens.
Background
The new ‘fragrance allergens’ labelling regulation (Commission Regulation (EU) 2023/1545 of 26 July 2023 amending Regulation (EC) No 1223/2009 of the European Parliament and of the Council as regards labelling of fragrance allergens in cosmetic products) aims to protect allergic individuals through providing them labelling information that allows them to make a proper choice when buying products. This Commission Regulation sets out new obligations for the labelling of an extended list of so called ‘fragrance allergens’ in addition to the one set by directive 2003/15/CE. The date of the entry into force of the new regulation is 16 August 2023. However, for new restrictions a transition period is foreseen until 31 July 2026 for placing products on the market, and until 31 July 2028 for withdrawal of products from the market.
At present, 24 so called ‘fragrance allergens’ (26 initially, but two are banned now) are to be listed in the list of ingredients even if they are constituents of a perfume, aroma or complex ingredient. Labelling is mandatory if the substance is present in the cosmetic product above specific threshold concentrations, which are different for leave-on and rinse-off products. The new regulation expands this list to 80+ allergens. Note that the purpose of this additional labelling is to inform those sensitised individuals who have been tested and know which ingredients to avoid. It will tell them whether the substance to which they are sensitised is present in the product. The formula has not changed. There is no requirement to remove these substances and no need to consider reformulating out of these ingredients. The overwhelming majority of cosmetic users will not experience any undesirable effects associated with the presence of these substances.
The expansion of the list of allergens introduced new regulatory and implementation challenges, such
as:
- Many complex names to be memorised by allergic consumers for the same type of allergen.
- Issue of space on labels due to very long list of ingredients.
To address these issues, a new annex III regulatory approach had to be developed in the new regulation, allowing substances with the same cross-sensitising properties to be listed under a common group name rather than the individual substance name.
A third challenge linked to the expansion of the list of allergens is the need to secure globally compatible labels. To reduce the impact at international level and facilitate the acceptance of the new EU labelling requirements globally, INCI names were allocated to those substances that had none and to the newly created group names. More details on the expected impacts are provided in the annex of the Cosmetics Europe guidance.
Purpose
The main purpose of this guidance document is to help companies to understand and correctly interpret the ‘fragrance allergens’ labelling requirements and adapt its practices to ensure continued compliance.
Sources of the 80+ allergens in cosmetic products
The term ‘fragrance allergens’ is a misnomer used to refer to contact allergens that are usually present in fragrance compositions. The new regulation itself uses the term ‘fragrance allergens’. Given that no definition is given to this term in the regulation, its use in the regulatory context could lead someone to think that the new labelling requirements apply only if the source of the allergen is in a fragrance mixture, which is not correct. The presence of the 80+ allergens is to be labelled regardless of its source.
An allergen can be present in a cosmetic product due to:
- Its presence in the fragrance or aroma mixture, either added directly or as part of a natural complex substance (e.g., essential oil, botanical extract).
- Its presence as a constituent of a Natural Complex Substance (NCS) (e.g., essential oil, botanical extract). At the Natural Complex Substance level, three cases shall be considered: The Natural Complex Substance may be identified as an allergen. A constituent of Natural Complex Substance may be identified as an allergen and a last case where both the Natural Complex Substance and its constituent may be identified as allergens. Note that in the case of an allergen that is constituent of another allergen (Natural Complex Substance), if their individual concentrations (considered separately) exceed the limit threshold, the legislation requires the label of both allergens - the natural complex substance and the constituent in question.
- Its presence as an impurity or contaminant. The unintended presence of one of the 80+ allergens, stemming from impurities of natural or synthetic ingredients, the manufacturing process, storage, or migration from packaging (although very unlikely) is to be mentioned in the list, if the concentration of the allergen in question exceeds the limit thresholds.
- A combination of two or more of the above (no contribution as a cosmetic ingredient). The Responsible Person needs to consider all the contributions of each of the sources and if the total concentration exceeds the limit threshold, the allergen is to be labelled.
- Direct use of the allergen as a cosmetic ingredient (i.e., not a fragrance or aroma mixture).
- Direct use of the allergen as a cosmetic ingredient and presence of the allergen as a combination of one or more of the above cases. Note that the direct use of the allergen as a cosmetic ingredient requires its labelling regardless of the concentration of the allergen in the product.
Order of declaration
According to article 19.1(g), ‘The list of ingredients shall be established in descending order of weight of the ingredients at the time they are added to the cosmetic product. Ingredients in concentrations of less than 1 % may be listed in any order after those in concentrations of more than 1 %.’
The listing of the presence of a substance is not part of the general ingredient listing. From a regulatory point of view, one can either apply the approach for ingredient listing (i.e. consider their concentration to define the position in the ingredient list) or put the allergens names at the end, since the allergens information is supplementary information to the ingredient list. The company can decide the approach they prefer. What would not be correct is to label the allergen in a random place in the ingredient list among those ingredients having a concentration >1%, as that would indicate a wrong concentration of the substance in the cosmetic product and can be considered as misleading the consumer.
Two approaches for naming of allergens: Standalone and Grouped
In the regulation there are two regulatory approaches for the naming of allergens in annex III, which are referred to as ‘Standalone’ and ‘Grouped’.
Standalone allergen entry approach
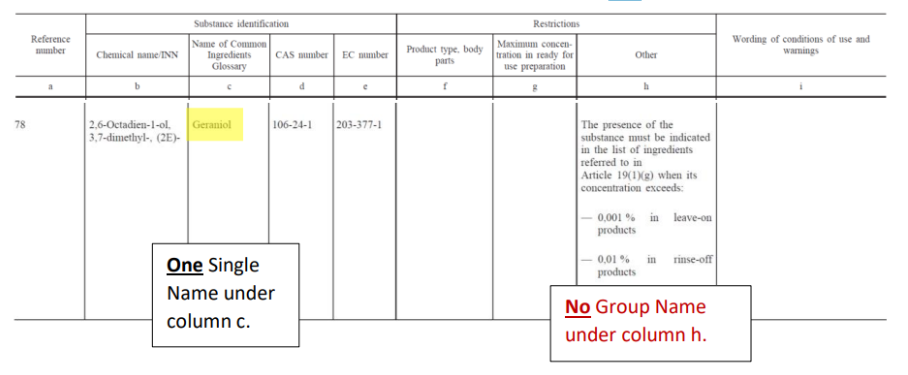
The Standalone allergen entry approach is the usual regulatory approach used in the past for the 24 allergens: i.e., one allergen = one substance = 1 Glossary/INCI name. In stand-alone allergen entries:
- the labelling requirement is set out by specifying the Limit Threshold under column h (‘Other’) and
- the labelling name to be used is mentioned under column c (‘Glossary’).
Example
For example (see Figure 1), if geraniol is present in the finished product in a concentration above 0.001% for leave-on and 0.01% for rinse-off, the ingredient has to be added to the list of ingredients as ‘geraniol’.
Figure 1: example of Standalone allergen entry – Geraniol.
Grouped allergen entry approach
The Grouped allergen entry approach is a new regulatory approach which was developed to solve the issue of the length of ingredient lists which would have made the label too long and complicated, and therefore not consumer friendly (especially for allergic people). Thus, instead of forcing the allergic person to memorise the whole list of substances belonging to the same cross-sensitisation group, only one name for each group is to be memorised by the concerned allergic consumers. That is the so called ‘Group Names’ (GN).
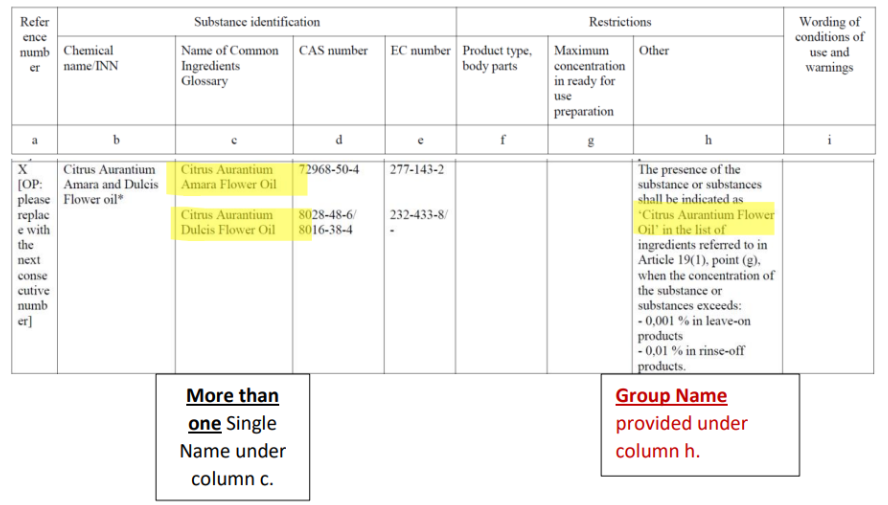
- The labelling name to be used to identify the allergen (Group Name) is specified under column h (‘Other’).
- The Single Names (INCI names) belonging to the same group are under column c (‘Glossary’).
It is important to outline that if the sum of the concentrations of the substances belonging to the same group is ABOVE the Limit Threshold, the use of the GN is mandatory. As regards Single Names (SN – i.e., the glossary name provided under column c of the entry) of the substance that belongs to the same group, its labelling in addition to the GN is not mandatory. A company can however decide to provide the SN as additional information, since the legislation does not forbid indicating additional information in the ingredient list.
However, if the sum of the concentrations of the substances belonging to the same group is BELOW the Limit Threshold, the use of the GN is NOT mandatory. In other words, a company can decide to use either the SN or the GN to fulfil the general labelling requirement for cosmetic ingredients (art. 19.1(g)). All GNs are INCI names. GNs could be used as synonym of SN.
Example
For example (see Figure 2), if a leave-on product contains 0,0008 % of citrus aurantium amara flower oil and 0,002 % of citrus aurantium dulcis flower oil, their sum exceeds the Limit Threshold for leave-on products. Thus, the two allergens are to be labelled by using the GN ‘Citrus aurantium flower oil’ (this is the ‘Group Name’, in the entry of Annex III provided under column h). If the company wish to also add the SNs ‘Citrus aurantium amara flower oil’ and ‘Citrus aurantium dulcis flower oil’ as additional information, that is possible, but it is not mandatory.
Figure 2: example of grouped allergens entry – Citrus Aurantium Flower Oil.
How do I recognise if the entry is a Standalone or Grouped allergen entry?
You need to look at whether a name (i.e., Group Name) is provided or not under column h:
- If column h specifies the name to be used, then that is a Grouped allergen entry,
- If column h does not specify the name, then that is a Standalone allergen entry.
Important notes
For the labelling of an allergen, the regulation states that it is the presence of the substance regardless of whether it has been added to a perfume mixture, NCS (Natural Complex Substance) or is present in other raw materials as cosmetic ingredient, that triggers the labelling, provided that the concentration exceeds the Labelling Threshold (LT) (i.e., 0,001 % in leave-on products or 0,01 % in rinse-off products). However, allergens that have been intentionally added as cosmetic ingredients shall be labeled regardless of whether their concentration exceeds the Labelling Threshold.
Lastly, for natural ingredients, the exact scope of an Annex III requirements is not always obvious from the wording of the entry. An example to illustrate this principle would be Cinnamomum cassia leaf oil. The labelling requirements laid down under column h (‘Other’) only apply to Cinnamomum cassia leaf oil. Other ingredients obtained from Cinnamomum cassia, such as Cinnamomum cassia extract, Cinnamomum cassia bark, Cinnamomum cassia bark powder, Cinnamomum cassia bark extract are out of the scope of the labelling requirements. Note, however, that ‘Cinnamomum cassia oil’ which does not specify the plant part from which the oil is obtained, would be considered as covered by the labelling requirement because it cannot be excluded that the oil was obtained from the leaves.
More information can be found in the Cosmetics Europe guidance.
